I recently discovered a consumer product known as Joulies. According to the Joulies Website, Joulies are "Engineered beans that cool coffee to the right temperature faster and keep it there longer". Sounds a little too good to be true...
I googled Joulies and was surprised to find that the testimonials I was reading were overwhelmingly positive. In addition, Joulies was on the popular TV show Shark Tank, and I couldn't imagine successfully lobbying a product that doesn't actually work. A final testament to Joulies credibility was the fact that Joulies are incredibly expensive. Of course, this is not something I like to admit, but at a cost of $60 for 5 little Joulies, I bought their story that Joulies are some sort of cutting edge solid state device that consumes latent heat at a critical melting point equal to the ideal drinking temperature of coffee, etc. etc.
So I bought some Joulies.
THE EXPERIMENT
Fortunately, I have been working on a different experiment regarding heat flow and have all the necessary equipment to put Joulies to the test. The goal is simple, I would like to test the claim made on the front page of the Joulies website:
I googled Joulies and was surprised to find that the testimonials I was reading were overwhelmingly positive. In addition, Joulies was on the popular TV show Shark Tank, and I couldn't imagine successfully lobbying a product that doesn't actually work. A final testament to Joulies credibility was the fact that Joulies are incredibly expensive. Of course, this is not something I like to admit, but at a cost of $60 for 5 little Joulies, I bought their story that Joulies are some sort of cutting edge solid state device that consumes latent heat at a critical melting point equal to the ideal drinking temperature of coffee, etc. etc.
So I bought some Joulies.
THE EXPERIMENT
Fortunately, I have been working on a different experiment regarding heat flow and have all the necessary equipment to put Joulies to the test. The goal is simple, I would like to test the claim made on the front page of the Joulies website:
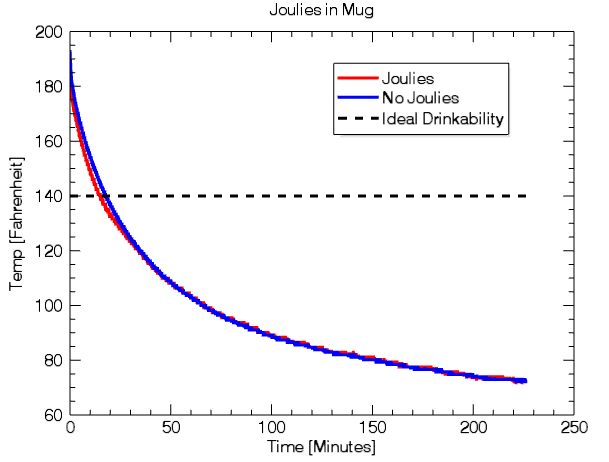
Namely, do Joulies cool hot liquid to 140 degrees and keep it at this temperature for hours?
For this system, convection is the dominant method of heat transfer (Newton's Law of Cooling), and the convective timescales dominate the conductive timescales which implies no spatial dependence to the temperature profile. Thus, the experiment is as simple as putting a temperature probe into the liquid and monitoring temperature as a function of time.
The equipment used to measure temperature were four Type K temperature sensors in combination with a PASCO Explorer-GLX. The liquid was hot water, and it was placed in identical ceramic mugs open to room temperature air.
There was only one trial, but all instruments and environmental factors were calibrated. The error in the measurements was less than 1 degree Fahrenheit and was primarily due to the GLX rounding decimals to integers before output.
RESULTS
For this system, convection is the dominant method of heat transfer (Newton's Law of Cooling), and the convective timescales dominate the conductive timescales which implies no spatial dependence to the temperature profile. Thus, the experiment is as simple as putting a temperature probe into the liquid and monitoring temperature as a function of time.
The equipment used to measure temperature were four Type K temperature sensors in combination with a PASCO Explorer-GLX. The liquid was hot water, and it was placed in identical ceramic mugs open to room temperature air.
There was only one trial, but all instruments and environmental factors were calibrated. The error in the measurements was less than 1 degree Fahrenheit and was primarily due to the GLX rounding decimals to integers before output.
RESULTS
If 140 degrees is the ideal drinkability for Joulies, then you reach this temperature 2 minutes and 41 seconds faster than without Joulies (14m57s compared to 17m38s).
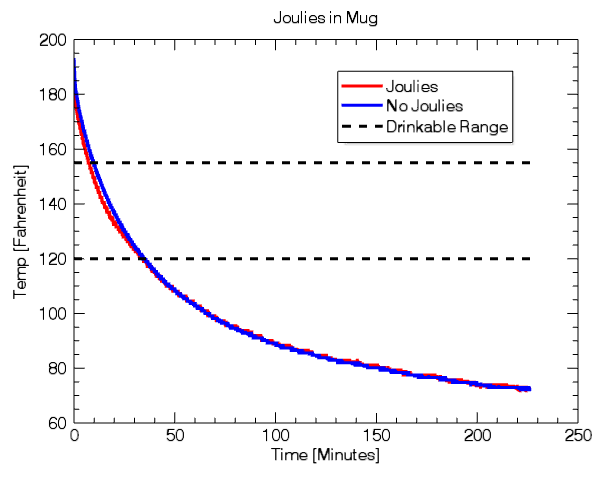
However, I think a more important question is how long before the liquid is cool enough to drink and how long does it stay at an enjoyable temperature? After burning my tongue a couple times, I discovered my personal preference for hot beverage temperature is between 120 and 155. At 155, it is hot enough to drink without fear of scalding, and at 120, it no longer has the pleasure associated with drinking a hot drink.
With this as our criteria for drinkability we find the following:
The Joulies reach enjoyable temperature in 7m6s and the control beverage reaches the same point 2m30s later.
However, there is absolutely no evidence that Joulies retain heat better than a typical ceramic mug. In fact, the ceramic mug without Joulies stays in the drinkable range for 20s longer than the mug with Joulies (34m21s compared to 34m01s).
CONCLUSION
Don't buy Joulies! Unless the two minute difference in cooling time is worth your money. Through clever marketing, Joulies has managed to create a successful company selling a virtually worthless product. I will be attempting to get my money back as soon as possible.
Joulies... more like FOOLIES!
However, there is absolutely no evidence that Joulies retain heat better than a typical ceramic mug. In fact, the ceramic mug without Joulies stays in the drinkable range for 20s longer than the mug with Joulies (34m21s compared to 34m01s).
CONCLUSION
Don't buy Joulies! Unless the two minute difference in cooling time is worth your money. Through clever marketing, Joulies has managed to create a successful company selling a virtually worthless product. I will be attempting to get my money back as soon as possible.
Joulies... more like FOOLIES!
UPDATE -- Joulies gave me my money back with no questions asked. Overall, their customer service was awesome. I completely forgot I dislike Joulies.



 RSS Feed
RSS Feed
